High-Efficiency Electrolyte for Li-Rich Cathode Materials Achieving Enhanced
Li-rich cathodes have been in considerable attention for their high reversible capacity. However, they have serious problems like poor cycling with intense capacity decay and voltage fading, which restrict their access to practical applications. In this work, a facile and efficient strategy is proposed to alleviate these intrinsic issues with a high-efficiency electrolyte system. This special electrolyte enables Li-rich cathodes to deliver superior integrated performance with a high initial discharge capacity of 301 mAh·g–1, outstanding cycling stability with a capacity retention of 88% at 0.5 C over 500 cycles, and a remarkable rate capability of 136 mAh·g–1 at 5 C, respectively. What is more, the voltage fading is largely suppressed. Physical and electrochemical characterizations demonstrate that the robust CEI film formed on the cathode surface contributes to the improved electrochemical performance. This work provides a new approach to surmount defects of Li-rich materials and will largely promote their practical applications on Li-ion batteries.
With the continuous implementation and promotion of the “double carbon” goal, electric vehicles, wind storage and other new energy industries have gradually become the current research hotspot. Lithium ion battery has always been the most widely used energy storage device. Improving the energy density of battery is one of the main directions of the development of lithium battery at present. The structure and composition of cathode material is an important factor affecting the energy density of battery.
The research group of advanced Energy Storage Materials and Technology led by Wu Jianfei, a researcher from Qingdao Energy Institute, has laid out a variety of lithium cathode material systems, and intensively studied the research and development of core high-energy density cathode materials such as high-nickel terpolymer and lithium-manganese rich base. In recent years, a series of breakthroughs have been made.
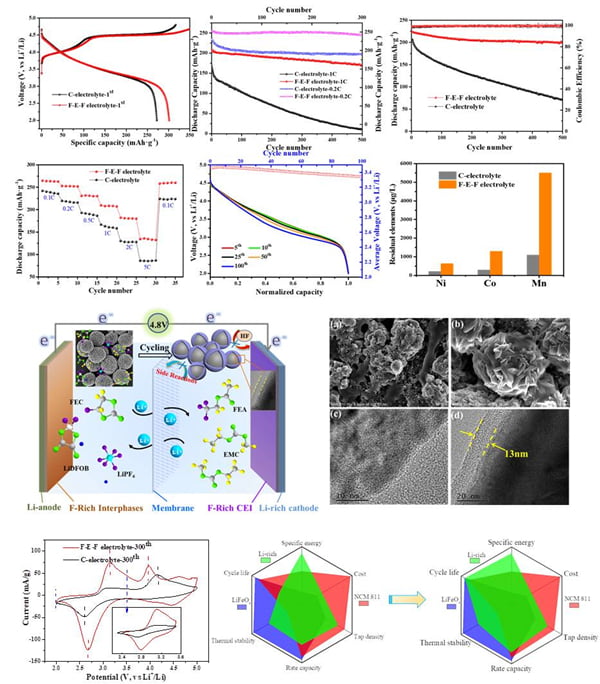
In the case of high-nickel terpolymer LiNi0.8Co0.15Al0.05O2 (NCA) cathode material, the research team successfully developed an alumina-coated magnesium doped NCA (Al2O3@Mg/NCA) system by an inside-out modification method. The NCA material constructed in this way has a stable internal structure. By inhibiting the phase transition and cationic doping inside the material, the rapid electron transmission inside the system can be achieved while inhibiting the occurrence of side reactions. At 1C, the battery went through 250 cycles and its capacity remained above 95%. This work lays a foundation for the commercialization of NCA materials, and also provides a new development idea for the design and modification of cathode materials for power batteries. With the increasing demand of high energy density battery for electric vehicles, the energy density of some cathode materials including NCA can no longer meet the demand, which severely limits the driving range of pure electric vehicles. Because of its high specific capacity, high energy density and low cost, lithium-manganese rich cathode material is very suitable for the new generation of high-energy density lithium-ion power batteries. However, its commercial application and development are restricted by its inherent defects such as low first efficiency, poor cycle and multiplier performance, serious voltage sag, and lack of suitable high voltage electrolyte. The research team successfully developed a full-performance lithium-manganese-rich battery system through one-step in-situ modification of electrolyte. The results were published in ACS Applied Materials & Interfaces. Through SEM/TEM/XPS/ICP-AES and other tests, it was confirmed that the solid and dense CEI protective film containing fluorine was in-situ coated on the surface of the positive and negative electrodes, which not only stabilized the structure of the positive electrode material, but also inhibited the corrosion of the positive electrode material caused by the decomposition of electrolyte, and effectively reduced the dissolution of metal ions. Thus, the comprehensive electrochemical performance of lithium – manganese – based cathode materials is greatly improved. The initial discharge capacity of the modified Li-rich cathode material reaches 300mAh·g-1, and the capacity retention rate is as high as 88% after 500 charge-discharge cycles at 0.5C rate. At the same time, the voltage drop during the battery cycle is also well improved, providing a new idea for the modification and commercial application of lithium-manganese based cathode materials (Figure 1).

FIG. 1. Excellent electrochemical performance was obtained by in-situ modification of lithium manganese based cathode material with one-step electrolyte.
As the main component of cathode material, cobalt has become one of the key factors restricting the supply chain of lithium ion battery industry due to its scarce resources and high price, which is also the biggest “bottleneck” problem facing the lithium battery industry at present. Low cobalt or even no cobalt has gradually become the trend of battery development. To this end, the research team innovatively developed a cobalt-free cathode material, which not only reduces the cost of the battery, but also improves the structural stability of the cathode material and the cycle stability performance of the battery, and further improves the overall electrochemical performance. The findings are published in ACS Applied Materials & Interfaces. Through SEM/HE-TEM/ in-situ XRD/XPS/CV characterization, it was found that cobalt-free cathode materials improved the structural degradation from lamellar to spinel and halite phases, and inhibited the structural deoxygenation and transition metal migration at high voltage during the cycle. The uniform and strong CEI and SEI films formed in situ on the surface of the positive and negative electrodes further enhance the structural stability of the positive and negative electrodes, and play an effective role in inhibiting the side reactions in the electrolyte, the occurrence of lithium dendrites and the corrosion of the positive and negative electrodes. Therefore, cobalt-free positive batteries show excellent cycling and rate performance: A high capacity retention rate of 99% and an ultra-low voltage drop of only 10mV can be achieved by using 0.5C multiplier cycle 200 times. Even if 1C high rate cycle 500 times still maintains 90% capacity, the recycled cathode material can still maintain a stable structure, the performance is in the leading level at home and abroad. This work achieved for the first time the “cobalt-free” of cathode materials with high specific energy, which improved the electrochemical performance and reduced the material cost of batteries. As a cathode material with great development prospects, it has great commercial value and application prospects (FIG. 2).
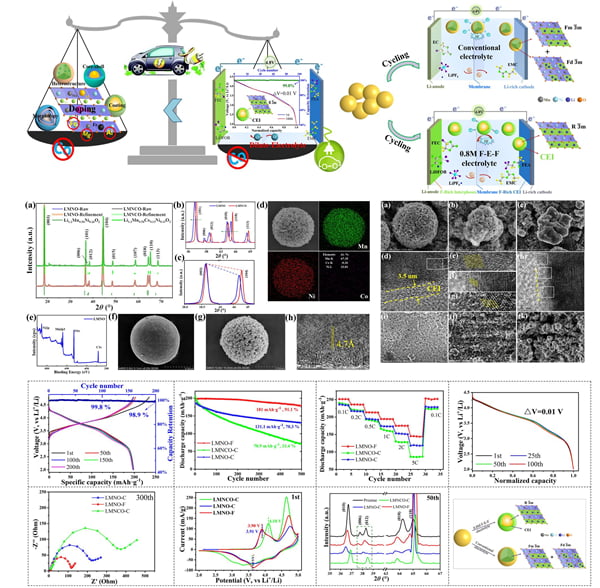
FIG. 2. The cathode material achieves high electrochemical performance by achieving cobalt-free oxidation.
In addition, the research team has presented a variety of solutions to existing problems in lithium battery and cathode materials, and has published a series of innovative research results (ACS Appl. Mater. Interfaces 2019, 11 15607; Adv. Mater. Interfaces, 2020, 2001698; Chemical Engineering Journal, 2022, 427, 131790).
The above work has been supported by two National Natural Science Foundation projects, two China Postdoctoral Science Foundation projects, two Natural Science Foundation projects of Shandong Province and key Research and Development Program projects of Shandong Province.
Link to original article:
https://pubs.acs.org/doi/10.1021/acsami.0c14995
Depeng Song, Jianfei Wu*, High-Efficiency Electrolyte for Li-Rich Cathode Materials Achieving Enhanced Cycle Stability and Suppressed Voltage Fading Capable of Practical Applications on a Li-Ion Battery. ACS Appl. Mater. Interfaces 2020, 12, 49666-49679.
https://pubs.acs.org/doi/full/10.1021/acsami.1c24580
Depeng Song, Jianfei Wu*, Dilute Electrolyte to Mitigate Capacity Decay and Voltage Fading of Co-Free Li-Rich Cathode for Next-Generation Li-Ion Batteries. ACS Appl. Mater. Interfaces. 2022, DOI: 10.1021/acsami.1c24580.